INTRODUCTION
After Gagner et al. [1] first introduced laparoscopic adrenalectomy for pheochromocytoma and Cushing syndrome in 1992, laparoscopic adrenalectomy has become the standard surgical procedure for adrenal tumors, replacing traditional open adrenalectomy. Laparoscopic adrenalectomy is known to have the advantages of fewer postoperative complications, bleeding, and analgesic requirements, a shorter hospital stay, and shorter time to return to normal physical activities and diet compared to open adrenalectomy [2,3]. Recently, indications for laparoscopic surgery have been expanded to include metastatic lesions and malignant tumors, and surgical safety for pheochromocytoma has been reported [4–6].
Mercan et al. [7] developed laparoscopic posterior retroperitoneal adrenalectomy (LPRA) in 1993 and LPRA has been used as one of the method of surgery for adrenal tumors until now. LPRA is a surgical method that accesses the adrenal gland through the back and has several advantages compared to laparoscopic transperitoneal adrenalectomy (LTPA); direct access to the adrenal gland, minimizing the need for intra-abdominal dissection, more useful for patient with previous abdominal surgery [8]. However, despite these advantages of LPRA, it is not as popular as LTPA because of the unfamiliar operative field and narrower working space and the worldwide experience has been limited [9–11]. Therefore, we report the initial experience of LPRA performed on 30 consecutive patients by one surgeon.
METHODS
Study population
From March 2018 to December 2019, a total of 30 consecutive patients diagnosed with adrenal tumor underwent surgical treatment at our hospital. We reviewed the medical records of these patients using the endocrine surgery database at Pusan National University Hospital. Various clinicopathologic features, including the age at operation, gender, body mass index (BMI), American Society of Anesthesiologist (ASA) grade, adrenal tumor size, length of hospital stay, type of disease, adrenal tumor site, estimated blood loss (EBL), mean operation time, operation type, perinephric fat density (FD), postoperative complication and postoperative pain were assessed. The perinephric FD was calculated as the ratio of the extent of retroperitoneal fat tissue and the area of retroperitoneal cavity using adrenal computed tomography. This study was approved by the Pusan National University Hospital Institutional Review Board (IRB No. J-2108-025-093). The informed consent was waived because this study design is a retrospective medical record review.
Operative technique
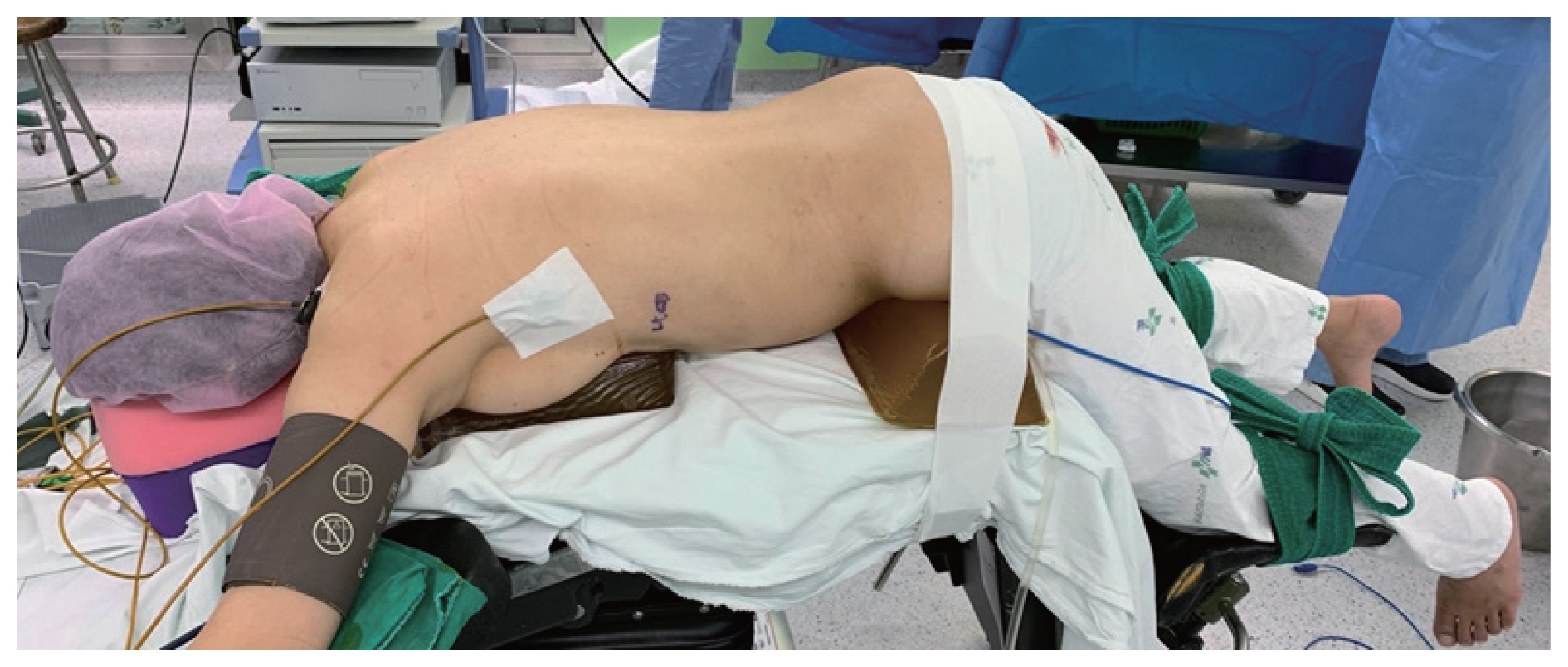
LPRA was performed with the patient in the prone position (Fig. 1). The first incision was made just below the tip of the 12th rib, and the retroperitoneal space was bluntly dissected with a finger. The second and third ports were then placed blindly on the finger (Fig. 2). After CO2 insufflation (15–20 mmHg), fatty tissue from the posterior aspect of the kidney was dissected and the superior pole of the kidney was exposed. Adrenalectomy was performed by resecting the adrenal gland from adjacent structures and ligating the adrenal vein. The resected adrenal gland was placed in an endo-bag and pulled out through the first incision site [11].
Statistical analysis
Data were expressed as mean±standard deviation or number (%) for descriptive statistics. A t-test was used for continuous variables. The Pearson product moment correlation chi-square test was used for categorical variables. In all cases, a P-value <0.05 was considered statistically significant. All statistical analyses were performed using IBM SPSS statistics 23.0 (IBM Corp., Armonk, NY, USA).
RESULTS
The clinicopathologic characteristics of the 30 patients are shown in Table 1. The mean age of the patients was 48 years and there were 13 males and 17 females. The mean BMI was 25.5 kg/m2 and 21 patients were grade II and nine patients were grade III in ASA grade. There was no bilateral adrenalectomy. All adrenalectomies were unilateral procedures. Primary hyperaldosteronism was the most frequently preoperative diagnosed disease (n=13, 43.4%), followed by adrenal incidentaloma (n=8, 26.6%), Cushing syndrome (n=5, 16.6%), and pheochromocytoma (n=4, 13.3%). In final pathologic result, adrenocortical adenoma was the most frequently operated disease (n=18, 60%), followed by pheochromocytoma (n=4, 13.3%), and adrenal hyperplasia (n=3, 10%). The mean size of postoperative adrenal tumor was 2.72 cm. The perioperative and postoperative various parameters of the 30 patients are shown in Table 2. The mean operating time was 162 minutes. Among the 30 patients, 28 patients underwent total adrenalectomy (93.3%) and two patients underwent cortical sparing adrenalectomy (6.7%). The mean perinephric FD was 0.4 cm and the mean postoperative adrenal weight was 29 g. The mean EBL during operation was 199 mL and the mean length of hospital stay was 3.7 days. In our study, maximal EBL was 1,500 mL when performing adrenalectomy with 5 cm-sized pheochromocytoma. There were no morbidity- or mortality-related complications in this study population. In only one patient, ilioinguinal neuralgia occurred after surgery and self-resolved within 1 week. There was no conversion to open adrenalectomy in this study. The mean day of postoperative analgesic intravenous injection with ketorolac tromethamine (Trolac) 30 mg was 1 day.
In our study, as the number of surgeries increased, the operating time became shorter and the operating time of last case (30th case) was 75 minutes (Fig. 3).
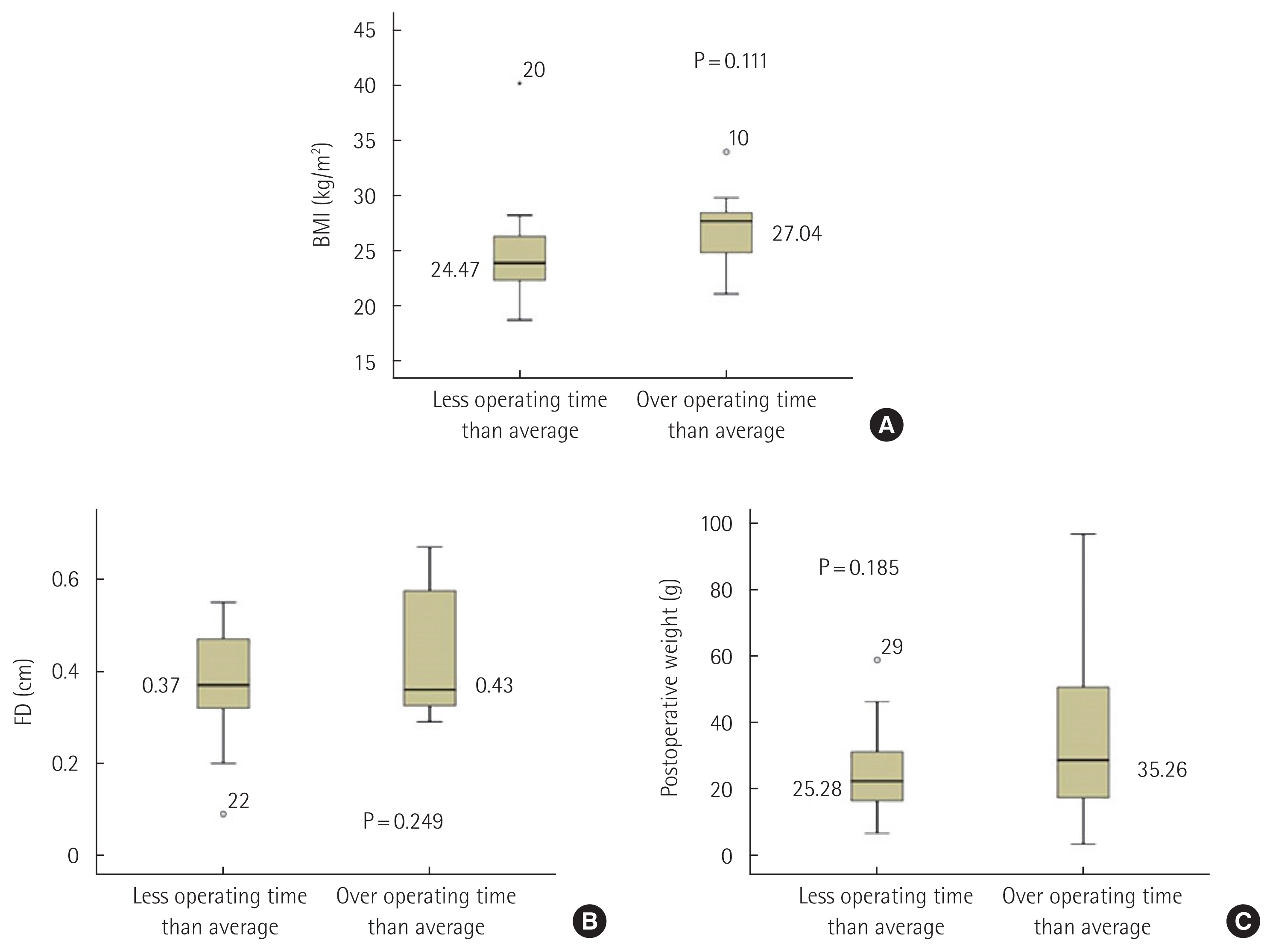
With analysis of the correlation between the various pre- and peri-operative parameters, we found with box plots that BMI (27.04 kg/m2 vs. 24.47 kg/m2, P=0.111) (Fig. 4A), perinephric FD (0.43 cm vs. 0.37 cm, P=0.249) (Fig. 4B) and postoperative adrenal weight (35.26 g vs. 25.28 g, P=0.185) (Fig. 4C) were higher in surgeries that took more time than the mean operating time compared to surgeries that took less time than the mean operating time with statistically insignificant. Box plots revealed that a wider range of perinephric FD and postoperative adrenal weight in surgeries with over operating time than average compared to surgeries with less operating time than average (Fig. 4B and C). We also found that when LPRA was performed for patients with BMI >23.16 kg/m2, the operating time was longer than the average (P=0.016).
DISCUSSION
Laparoscopic adrenalectomy is considered as the gold standard surgical method for removing benign adrenal mass [12,13]. Various studies described the advantage of laparoscopic adrenalectomy [12,14] and LTPA has been most popular surgical procedure for adrenal tumor, except in some special cases, such as primary malignant tumor or metastatic malignant tumor. After Mercan et al. [7] introduced LPRA in 1993, LPRA has recently been widely used as a surgical method for resection of adrenal tumors. Various previous studies reported the differences between LTPA and LPRA. In several studies, no significant differences in outcome variables were found between the LPRA versus the classic LTPA [15–17]. However, recent studies reported shorter hospital stay and lower postoperative pain for LPRA than for LTPA [18,19]. In this study, we reported no complications after LPRA, except one case with ilioinguinal neuralgia. The location of the adrenal glands in the retroperitoneal space and the direct approach explains the shorter operation times and lower amount of blood loss because of minimized intra-abdominal dissection in LPRA compared to LTA [11]. The adrenal gland is exposed directly in the retroperitoneal space, without mobilization of liver, pancreas and spleen in LPRA, reflecting low complication rates. Our complication, ilioinguinal neuralgia, is a complication that occurs not only in LPRA but also in laparoscopic surgery and improves after a few days.
LPRA could be also performed for patients with previous abdominal surgery. Furthermore, it is performed in case of bilateral adrenal resection without the need for repositioning and is feasible in obese patients as the abdominal fat is located at the non-operative ventral side of the patient. However, because most surgeons are not familiar with the anatomy of the retroperitoneal space, a substantial learning time was required for the use of LPRA [11,20]. Previous study suggested that about 20–25 LPRAs would probably be necessary to apprehend the new technique for LPRA [21]. In our study, the operating time of first case was 330 minutes, but, as the number of surgeries increased, the operating time became shorter and the operating time of last case (30th case) was 75 minutes.
In our study, the maximal preoperative and postoperative tumor size was 20 cm and 8.2 cm, respectively (Table 1). In that case, adrenal tumor was a 20 cm-sized pure adrenal cyst and LPRA was performed while the adrenal cystic fluid was aspirated. Walz et al. [22] showed that the retroperitoneal approach is difficult to perform in patients with large tumors (>7–8 cm), but we think that LPRA is suitable and safe for patients with over 7–8 cm-sized benign pure adrenal cyst. Furthermore, maximal EBL was 1,500 mL when performing adrenalectomy with 5cm sized pheochromocytoma (Table 2). During performing adrenalectomy for pheochromocytoma, the tumor often bleeds, and once the tumor begins to bleed, it is often out of control. In this case, we think that it is important to control bleeding by gauze packing unless it is a major blood vessel injury. If electrocautery is excessively performed on the tumor bleeding site, bleeding may be further promoted.
In our study, despite statistically insignificant value, BMI, retroperitoneal FD and postoperative adrenal weight had correlations with the operating time. Walz et al. [22] showed that the retroperitoneal approach is difficult to perform in patients with a high BMI. However, more present study showed no correlation between BMI and the outcome measures (operation time, recovery time, and blood loss) [11]. Therefore, BMI, retroperitoneal FD and postoperative adrenal weight were not impossible factors in implementing LPRA, but they may be factors that should be sufficiently considered when performing LPRA.
In conclusion, based on our initial experiences for LPRA in single tertiary center, LPRA was suitable and safe for patients with benign adrenal tumors. BMI, retroperitoneal FD and postoperative adrenal weight may be related to the operating time, so they should be considered when deciding on a surgical method for adrenalectomy.