INTRODUCTION
In breast cancer patients, axillary lymph node (LN) status is considered the most important predictor of survival in the absence of distant metastasis [1,2]. Also, the need for and selection of various local or systemic adjuvant therapies are determined according to the extent of involvement of axillary LNs, and the decision of radiation therapy and fields of treatment should be based upon the pretreatment stages in patients treated with neoadjuvant chemotherapy [3]. Non-invasive imaging techniques, including ultrasonography (US), computed tomography, magnetic resonance imaging (MRI), and positron emission tomography-computed tomography, are being used for the purpose of preoperative axillary nodal staging. Among these, axillary US has been demonstrated to be the most accurate and sensitive method. Its accuracy and sensitivity can be further increased if any ultrasonographically suspicious or indeterminate LNs are also subjected to ultrasound-guided fine needle aspiration cytology (FNAC) or core needle biopsy (CNB) [4-14]. Although ultrasonographic criteria used to identify abnormal nodes have been previously described [8,9], there are few studies applying these criteria to determine the number of metastatic LNs. On the basis of the assumption that the progressive pattern of tumor involvement in the axillary nodes can be reliably reproduced by US, we attempted to compare the preoperative ultrasonographic findings between pN1 and pN2-3 stages in breast cancer patients with pathologically-confirmed axillary LN metastases.
METHODS
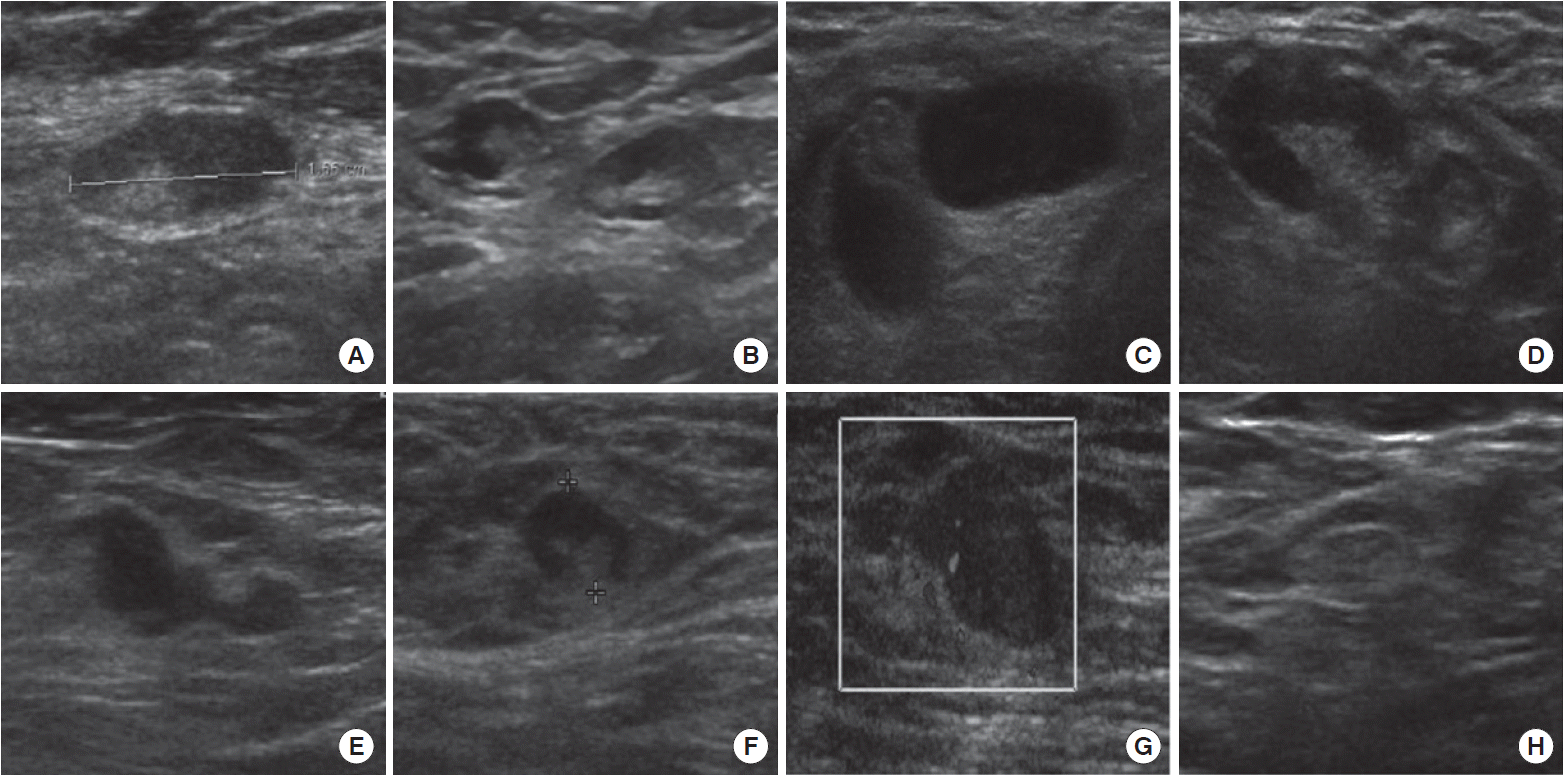
We performed a retrospective study of consecutive 140 patients with histopathologically confirmed axillary LN metastases who were diagnosed with invasive breast cancer, and underwent operation without preoperative chemotherapy or endocrine therapy at our institution between January 2004 and June 2008. The institutional review board approved this study and waived the requirement for informed consent. All patients were surveyed by axillary US in the outpatient clinic, either at the time of the initial evaluation of the breast primary, or as part of the first visit to our hospital for referred patients. Static axillary LN ultrasonographic images were classified retrospectively by an experienced radiologist into the following 8 categories [8-13]: (A) eccentric cortical thickening/lobulations, (B) mixture of normal and abnormal nodes, (C) complete loss of central echogenicity or fatty hilum, (D) markedly hypoechoic cortex, (E) abnormally roundeded or irregular shape, (F) increased peripheral blood flow, (G) compression of the hyperechoic medullary region, and (H) normal node (Fig. 1). All patients were divided into pN1 and pN2-3 stages according to the number of metastatic axillary LNs, and then we analyzed whether or not their ultrasonographic findings are different between the two groups. Statistical analyses were performed using SPSS ver. 15.0 (SPSS Inc., Chicago, IL, USA). The differences in ultrasonographic abnormalities between pN1 and pN2-3 stages were analyzed by chi-squared test, and the mean number of metastatic LNs according to sonographic abnormalities was compared by one-way ANOVA. A P<0.05 was considered statistically significant. For the pathological staging of the axilla, this study used the guidelines and terminology proposed by the 7th edition of the American Joint Committee on Cancer staging manual [15].
RESULTS
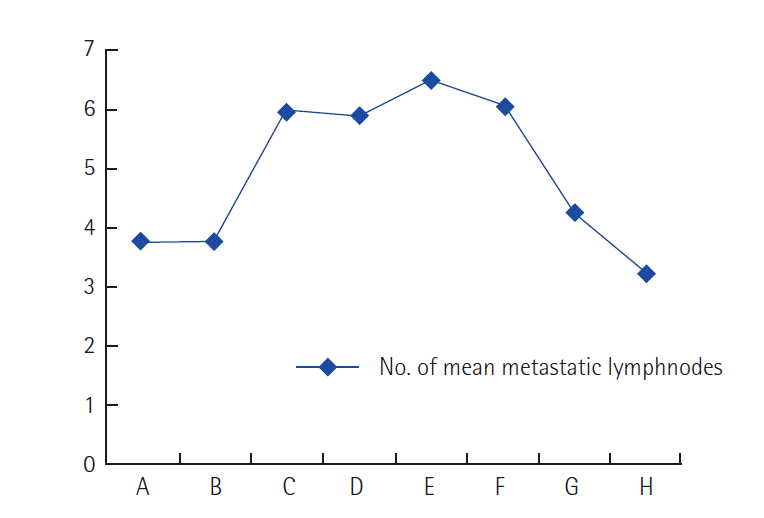
One hundred and forty patients (mean age, 47.5±10.6 years) who underwent a preoperative US were reviewed. The primary tumor was diagnosed in 109 patients (77.9%) by CNB, and 19 patients (13.6%) by FNAC, 7 patients (7.9%) by frozen section, and 1 patient by vacuum-assisted biopsy (VAB). The mean tumor size was 2.6± 1.4 cm (range, 0.4 to 13.5 cm) and mean retrieved LN number was 17.7±7.4 (range, 6 to 48), the mean metastatic LN number was 4.5±4.8 (range, 1 to 31), and the mean lymph node ratio, expressed as the ratio of positive excised nodes, was 0.26±0.24 (0.04 to 1). Modified radical mastectomy was performed in 116 patients, including 14 transverse rectus abdominis myocutaneous flap and 2 lattismus dorsi flap reconstructions. Wide excision with axillary lymph node dissection was performed in 24 patients (Table 1). The order of frequency of ultrasonographic abnormalities in axillary nodes were “eccentric cortical thickening/lobulation” (43.6%), followed by “markedly hypoechoic cortex” (30.0%), “complete loss of central echogenicity/fatty hilum” (29.3%) and “abnormally rounded/ irregular shape” (22.1%) and the frequency of normal nodes was 26.4% (Table 2). The pathologic axillary nodal stages were N1 in 83 patients (59.3%) and N2-3 in 57 patients (40.7%) (Table 1). In the 8 categories of ultrasonographic findings, eccentric cortical thickening/lobulations (63.9%), mixture of normal and abnormal nodes (73.3%) and compression of the hyperechoic central medullary region (51.9%), and normal nodes (67.6%) were higher in pN1 than pN2-3 stages, but not with statistical significance (Fig. 2, Table 2). In contrast, complete loss of central echogenicity/fatty hilum, markedly hypoechoic cortex, abnormally rounded/irregular shape, and increased peripheral blood flow were higher in pN2-3 than pN1 stages (Fig. 2, Table 2). Among these, complete loss of central echogenicity/fatty hilum, and abnormally rounded/irregular shape are significantly higher in pN2-3 stages. Odds ratios were 2.443 and 2.485, respectively (Table 2). The number of mean metastatic LNs is statistically significantly greater in patients with abnormally round/irregular shape (P=0.021) (Table 2, Fig. 3).
DISCUSSION
Axillary node dissection is performed as part of the surgical treatment of breast cancer, mainly for staging, and for planning adjuvant therapy according to the number of metastatic LNs [3,16]. Currently, sentinel LN mapping and resection in the surgical staging of the axilla is recommended as the preferred method to assess the pathologic status of axillary LN in patients with clinically node-negative breast cancer and those with a negative sentinel node considered equivalent to disease-free axilla [3,17,18]. However, sentinel node biopsy is not recommended to assess the pathologic status of the axillary LN in patients with inoperable, locally advanced breast cancer and after preoperative chemotherapy.
Also, the initial use of anthracycline-based preoperative chemotherapy with or without taxane, which has proven its superior efficacy, is standard therapy [3,19,20] and radiation therapy should be performed to reduce locoregional recurrence after mastectomy in initial clinical locally advanced breast cancer patients regardless of the response of preoperative chemotherapy [3,21]. For autologous tissue reconstruction, delayed reconstruction is generally preferred after completion of radiation therapy due to loss of reconstruction cosmesis [3,22]. However, since preoperative treatment may eradicate the foci of disease in axillary LNs, the potential loss of long-term clinical prognostic information may complicate clinical decision making for local or systemic therapy [17,23]. For this reason, an innocuous and inexpensive, nonsurgical diagnostic procedure that could reliably predict the extent of axillary nodal involvement preoperatively would be extremely valuable.
The purpose of this study was to demonstrate the clinical advantages that could be gained by the application of preoperative axillary US in breast cancer patients, especially locally-advanced breast cancer, and tried to present the most useful characteristics of ultrasonographic findings to preoperatively evaluate axillary nodal status in these patients. US has been proven to have higher sensitivity (62%-72%) and specificity (69%-95%) than clinical examination and other imaging techniques in the assessment of axillary LN involvements [4-8].
Anatomically, LN has a cortex that contains primary or secondary follicles and a paracortex and medulla that contains the sinuses, lymphoid tissue, and variable amounts of fat and vessels. In US, normal LN is represented as oval-shaped structure composed of an outer hypoechoic cortex and an inner hyperechoic medulla [24]. The process of LN metastasis is represented as tumor cells initially seen as clusters in afferent lymphatic vessels, and which, in the peripheral sinuses, gradually spread to the medulla, replace the entire LN, and extend into adjacent extranodal tissues [25]. Therefore, in early-stage breast carcinoma, tumor deposits essentially restricted within the LN cortex represent either as microscopic nodules with little to no desmoplastic response or as a thin layer of tumor cells directly beneath the LN capsule or within the subcapsular sinuses, whereas in patients with advanced nodal disease, complete nodal replacement and true extracapsular extension are more frequently present [13]. These progressive patterns of tumor involvement in axillary nodes in patients with breast carcinoma can be to some extent reproduced by ultrasonography because of the ability of this technique to show the different anatomic nodal compartments and their respective alterations [24]. The early metastatic features are sonographically represented by asymmetric thickening and focal lobulations of the cortex, asymmetric compression of hyperechoic mediastinum, whereas late histological features are represented by a complete loss of the medullary or fatty hilum, markedly decreased echogenicity of the entire node, and mediastinal displacement, as well as necrosis and accompanying changes in the shape of the LN, including abnormally rounded or spiculated contours [13,22,26,27].
In our study, the common frequency of eccentric cortical thickening/ lobulations and the relatively high findings of normal nodes are thought to be due to the fact that early metastatic diseases, corresponding to the N1 stage, comprised about 60% of our patients (Table 1, Fig. 2). In patients whose ultrasonographic findings suggested complete nodal replacement or extracapsular extension, such as complete loss of central echogenicity or fatty hilum, and abnormally rounded or irregular shape, had a higher burden of metastatic disease in their axillae (≥4 positive nodes) than patients whose ultrasonographic findings were normal, or suggested to be confined to the cortex, such as eccentric cortical thickening/lobulations (Table 2). These results are consistent with other studies indicating that massively involved LNs are easily recognized as rounded or irregularly shaped masses [13,26,27].
Therefore, in our opinion, if ultrasonographic findings such as complete loss of central echogenicity/fatty hilum or abnormally rounded/irregular shape exist in preoperative axillary US, these patients could forgo sentinel node mapping, and initial use of preoperative chemotherapy should be actively considered. Also, post-mastectomy radiation therapy to the chest wall and axilla including supraclavicular area should be strongly considered due to the possibility of axillary down-staging after neoadjuvant therapy in these patients, and when autologous tissue reconstruction is considered, delayed reconstruction seems to be the best way to avoid loss in reconstruction cosmesis. Furthermore, comparing pathologic and clinical nodal stages by using axillary US might be helpful in the response evaluation of axillary LNs after neoadjuvant therapy.
We have suggested that preoperative axillary US can indicate the process of tumor involvement in axillary LN, and to some degree predict the extent of axillary nodal involvement. Patients whose ultrasonographic surveys show complete loss of central echogenicity/fatty hilum, or abnormally rounded/irregular shape, are most likely to have a higher burden of metastatic disease in their axillae (≥4 positive nodes), corresponding to the pN2-3 axillary stage. Classifications of preoperative ultrasonographic findings of axillary LN may help provide more exact pretreatment clinical axillary nodal staging, especially in a neo-adjuvant setting.